In many ways, biopharmaceutical products have revolutionized the medical industry. They allow us to prevent and treat diseases that would once have been a lifelong burden, or even a death sentence. However, they are still new enough that many of us still have unanswered questions. For example, what is the difference between biologics and biosimilars? Let’s talk about it.
What are Biologics?
Biologics are drug products that are manufactured using materials from living sources. There are a few common types of biologics, including extracted (taken directly from a living system), semi-synthesized (produced with recombinant DNA technology), vaccines, and gene therapies.
What are Biosimilars?
A biosimilar is a biologic that is similar enough to an existing FDA-approved product that it performs in exactly the same way, but different enough to avoid legal battles with the producers of the original product. Essentially, a biosimilar is a generic version of a biologic.
Biologics vs. Biosimilars
Biologics often improve patient outcomes by a great deal, but they can also be cost-prohibitive. That’s where biosimilars come in. Biosimilars are just as rigorously tested as biologics, and offer all of the same benefits, clinically speaking—they are equally safe and effective. The only meaningful difference between the two is usually price. Because biosimilars have a more streamlined development process than biologics, they are typically more affordable.
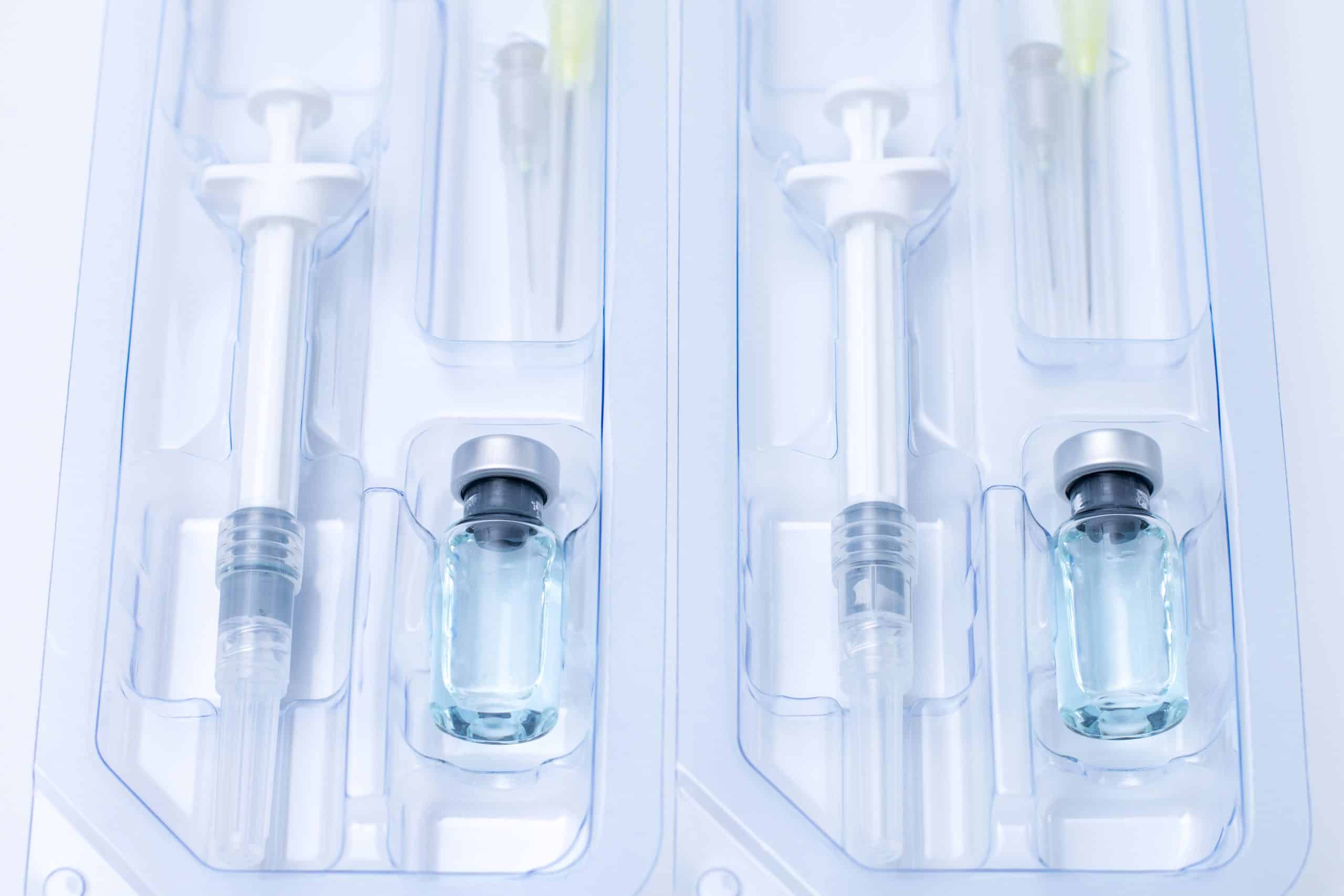
Covid-19, flu, coronavirus liquid vaccine vial bottle and syringe set in plastic package container preparing for injection on white background. Vaccination for fight Covid-19 virus concept.
Biopharma Packaging from Tjoapack
One thing biologics and biosimilars have in common is that they both require high-quality packaging. At Tjoapack, we provide a range of top of the line biopharma packaging solutions. For more information, contact us today. Call (865) 494-6000 or send an email to solutions@tjoapack.com.